Classified by application
All Products
Signaling Pathways
- Angiogenesis/Protein Tyrosine Kinase
- Apoptosis Pathway
- Cell Cycle/DNA Damage
- Epigenetics
-
GPCR/G Protein
- Urotensin Receptor
- Melatonin Receptor
- Neurotensin Receptor
- Glucose Transporter
- Motilin Receptor
- TGR5
- Cholecystokinin Receptor
- Adenylate Cyclase
- Somatostatin Receptor
- Oxytocin Receptor
- Bradykinin Receptor
- Guanylate Cyclase
- Melanocortin Receptor
- Imidazoline Receptor
- Neuropeptide Receptor
- PAR
- CRFR
- Sigma Receptor
- S1P Receptor
- GHSR
- PKA
- OX Receptor
- Adenosine Receptor
- Glucagon Receptor
- Prostaglandin Receptor
- NK Receptor
- GnRH Receptor
- Leukotriene Receptor
- Glucocorticoid Receptor
- mAChR
- P2 Receptor
- GPR
- CGRP Receptor
- CXCR
- Histamine Receptor
- CaSR
- Ras/Rho
- Vasopressin Receptor
- Smoothened/Smo
- mGluR
- Opioid Receptor
- Dopamine Receptor
- Cannabinoid Receptor
- Angiotensin Receptor
- Adrenergic Receptor
- SGLT
- Endothelin Receptor
- CCR
- 5 HT Receptor/Serotonin Receptor
- Hormone Pathay
- Ion Channel/Membrane Transporter
- Jak/Stat Pathway
- MAPK Pathway
- Microbiology/Virology
-
Neuro Signaling Pathway
- Imidazoline Receptor
- Monoamine Oxidase
- COMT
- Amyloid β
- NK Receptor
- Beta secretase
- AChE
- SSRI
- CaMK
- P glycoprotein
- Opioid Receptor
- Dopamine Receptor
- Adrenergic Receptor
- Histamine Receptor
- mGluR
- Gamma secretase
- Dopamine Transporter
- 5 HT Receptor/Serotonin Receptor
- P2 Receptor
- FAAH
- CGRP Receptor
- nAChR
- COX
- GABA Receptor
- mAChR
- NFκB
- PI3K/Akt/mTOR
-
Protease/Metabolic Enzyme
- COMT
- 11β HSD
- Carboxypeptidase
- ACC
- Xanthine Oxidase
- Pyruvate Kinase
- Mitochondrial Metabolism
- ALDH
- SCD
- Serine Protease
- MLR
- FTase
- CETP
- Neprilysin
- MAGL
- Carbonic Anhydrase
- E1 E2 E3 Enzyme
- Elastase
- PAI1
- NOX
- ROR
- Glucokinase
- Aldose Reductase
- Tyrosinase
- FAS
- DGAT
- Lipoxygenase
- Nampt
- IDO
- IDH
- MMP
- Cathepsin
- Phosphatase
- Thrombin
- FXR
- RAR/RXR
- HMGCR
- FAAH
- Gamma secretase
- RAAS/ACE
- Caspase
- HIV Protease
- Aminopeptidase
- HCV Protease
- HSP
- Tryptophan Hydroxylase/TPH
- Factor Xa
- DPP
- Procollagen C Proteinase
- Integrase
- Phosphodiesterase/PDE
- Phospholipase
- LXR
- Proteasome
- Cytochrome P450
- TGF beta/Smad
- Wnt/Stem Cell
- Others Pathway
Research Areas
- Metabolic Disease
- Neurological Disease
- Endocrinology
- Diabetes
- Cardiovascular Disease
- Immunology
- Allergy
- Inflammation
- Infection
- Cancer
Nature products
Antibodies
Peptides
Catalysts
Impurities
Intermediate
- Amino acid
- Ammonia Series
- Azaindazole
- Pyridazine Series
- Nucleoside Series
- Quinazoline
- Pyrazole Series
- Boric acid
- Pyrimidine Series
- Quinoline Series
- Pyrrole Series
- Thiophene Series
- Saccharide
- Azaindole
- Indole Series
- Piperidine Series
- Indazole Series
- Heterocyclic Series
- Benzene Series
- Pyridine Series
Raw Materials
PDK
| Chemical Structure | Cat. No. | Product Name | CAS No. |
|---|
 |
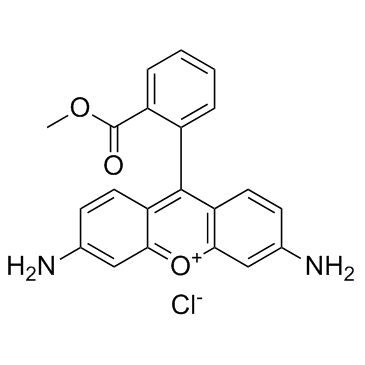
BCP37870 | PS210 New | 1221962-86-2 |
PS210 is a substrate-selective inhibitor of protein kinase PDK1.
|
|||
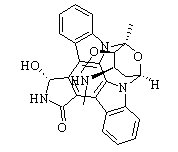 |
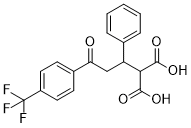
BCP14345 | UCN-01 New | 112953-11-4 |
UCN-01 (7-hydroxystaurosporine) acts as an inhibitor against many phosphokinases, such as the serine/threonine kinase AKT, calcium-dependent protein kinase C, and cyclin-dependent kinases.
|
|||
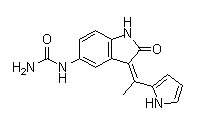 |
BCP16225 | PDK1 inhibitor2 New | 850717-64-5 |
BX517 is a potent and selective inhibitor of PDK1 with IC50 of 6 nM.
|
|||
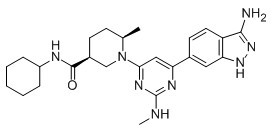 |
BCP08386 | GSK 2334470 | 1227911-45-6 |
Potent 3-phosphoinositide-dependent protein kinase (PDK1) inhibitor (IC50 ~ 10 nM).
|
|||
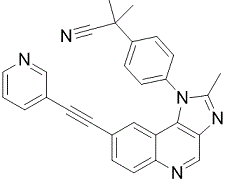 |
BCP01894 | BAG956 | 853910-02-8 |
NVP-BAG956(BAG 956) is a potent, ATP-competitive and selective dual PI3K and PDK1 inhibitor in vitro and in vivo,with IC50 values to be 56, 444, 34, 117 and 240 nM for PI3K p110 alpha, beta, delta and gamma and PDK1 kinases, respectively.
|
|||
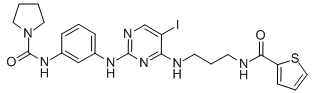 |
BCP01899 | BX795 | 702675-74-9 |
BX795 is shown to be a specific and potent inhibitor of IκB kinase ε (IKKε), phosphoinositide 3-kinase 1 (PDK1), and TANK-binding kinase 1 (TBK1).
|
|||
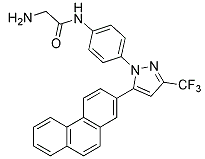 |
BCP01835 | OSU-03012 | 742112-33-0 |
OSU03012; also known as AR12, is an orally available, targeted anti-cancer agent that has been shown in pre-clinical studies to inhibit PDK-1,OSU-03012 is a recently licensed novel derivative of the cyclooxygenase-2 (COX-2) inhibitor celecoxib .
|
|||
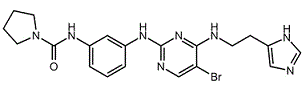 |
BCP01898 | BX-912 | 702674-56-4 |
BX-912 is a inhibitor of 3-Phosphoinositide-dependent Kinase-1 (PDK1).
|
|||
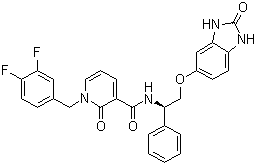 |
BCP06483 | PDK1 inhibitor New | 1001409-50-2 |
PDK1 inhibitor is a phosphoinositide-dependent kinase-1 (PDK1) inhibitor.
|
|||
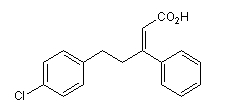 |
BCP08452 | PS 48 | 1180676-32-7 |
Phosphoinositide-dependent protein kinase-1 (PDK1) activator (Kd = 10.3 μM). Binds exclusively to the PIF-binding pocket of PDK1, distinct from the ATP binding site.
|
|||