Classified by application
All Products
Signaling Pathways
- Angiogenesis/Protein Tyrosine Kinase
- Apoptosis Pathway
- Cell Cycle/DNA Damage
- Epigenetics
-
GPCR/G Protein
- Urotensin Receptor
- Melatonin Receptor
- Neurotensin Receptor
- Glucose Transporter
- Motilin Receptor
- TGR5
- Cholecystokinin Receptor
- Adenylate Cyclase
- Somatostatin Receptor
- Oxytocin Receptor
- Bradykinin Receptor
- Guanylate Cyclase
- Melanocortin Receptor
- Imidazoline Receptor
- Neuropeptide Receptor
- PAR
- CRFR
- Sigma Receptor
- S1P Receptor
- GHSR
- PKA
- OX Receptor
- Adenosine Receptor
- Glucagon Receptor
- Prostaglandin Receptor
- NK Receptor
- GnRH Receptor
- Leukotriene Receptor
- Glucocorticoid Receptor
- mAChR
- P2 Receptor
- GPR
- CGRP Receptor
- CXCR
- Histamine Receptor
- CaSR
- Ras/Rho
- Vasopressin Receptor
- Smoothened/Smo
- mGluR
- Opioid Receptor
- Dopamine Receptor
- Cannabinoid Receptor
- Angiotensin Receptor
- Adrenergic Receptor
- SGLT
- Endothelin Receptor
- CCR
- 5 HT Receptor/Serotonin Receptor
- Hormone Pathay
- Ion Channel/Membrane Transporter
- Jak/Stat Pathway
- MAPK Pathway
- Microbiology/Virology
-
Neuro Signaling Pathway
- Imidazoline Receptor
- Monoamine Oxidase
- COMT
- Amyloid β
- NK Receptor
- Beta secretase
- AChE
- SSRI
- CaMK
- P glycoprotein
- Opioid Receptor
- Dopamine Receptor
- Adrenergic Receptor
- Histamine Receptor
- mGluR
- Gamma secretase
- Dopamine Transporter
- 5 HT Receptor/Serotonin Receptor
- P2 Receptor
- FAAH
- CGRP Receptor
- nAChR
- COX
- GABA Receptor
- mAChR
- NFκB
- PI3K/Akt/mTOR
-
Protease/Metabolic Enzyme
- COMT
- 11β HSD
- Carboxypeptidase
- ACC
- Xanthine Oxidase
- Pyruvate Kinase
- Mitochondrial Metabolism
- ALDH
- SCD
- Serine Protease
- MLR
- FTase
- CETP
- Neprilysin
- MAGL
- Carbonic Anhydrase
- E1 E2 E3 Enzyme
- Elastase
- PAI1
- NOX
- ROR
- Glucokinase
- Aldose Reductase
- Tyrosinase
- FAS
- DGAT
- Lipoxygenase
- Nampt
- IDO
- IDH
- MMP
- Cathepsin
- Phosphatase
- Thrombin
- FXR
- RAR/RXR
- HMGCR
- FAAH
- Gamma secretase
- RAAS/ACE
- Caspase
- HIV Protease
- Aminopeptidase
- HCV Protease
- HSP
- Tryptophan Hydroxylase/TPH
- Factor Xa
- DPP
- Procollagen C Proteinase
- Integrase
- Phosphodiesterase/PDE
- Phospholipase
- LXR
- Proteasome
- Cytochrome P450
- TGF beta/Smad
- Wnt/Stem Cell
- Others Pathway
Research Areas
- Metabolic Disease
- Neurological Disease
- Endocrinology
- Diabetes
- Cardiovascular Disease
- Immunology
- Allergy
- Inflammation
- Infection
- Cancer
Nature products
Antibodies
Peptides
Catalysts
Impurities
Intermediate
- Amino acid
- Ammonia Series
- Azaindazole
- Pyridazine Series
- Nucleoside Series
- Quinazoline
- Pyrazole Series
- Boric acid
- Pyrimidine Series
- Quinoline Series
- Pyrrole Series
- Thiophene Series
- Saccharide
- Azaindole
- Indole Series
- Piperidine Series
- Indazole Series
- Heterocyclic Series
- Benzene Series
- Pyridine Series
Raw Materials
Impurities
| Chemical Structure | Cat. No. | Product Name | CAS No. |
|---|
 |
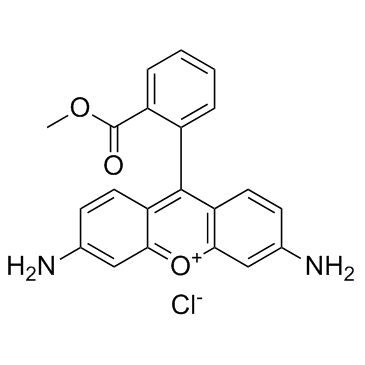
BCP49647 | Peramivir Impurity 05 New | 383910-22-3 |
 |
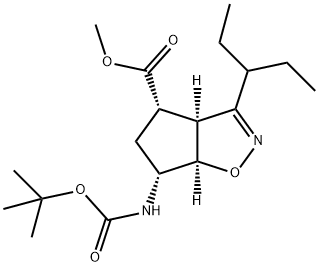
BCP49588 | Landiolol Hydrochloride EnantiomerⅢ New | 1253907-81-1 |
 |
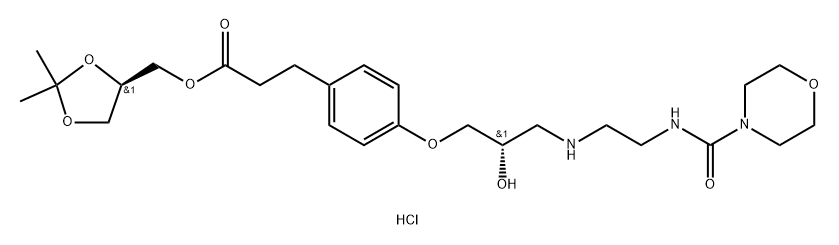
BCP49587 | Paliperidone Formylation Impurity New | 1204315-39-8 |
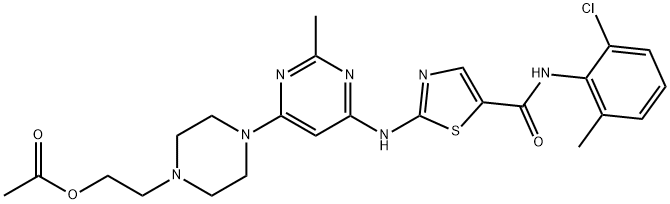 |
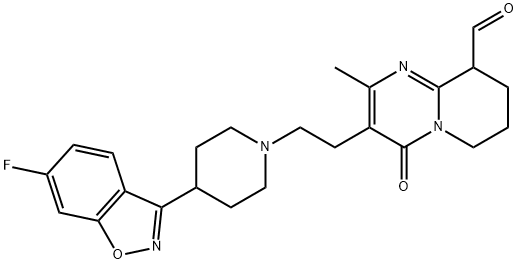
BCP49581 | Dasatinib Impurity 5 New | 1245157-85-0 |
 |
BCP49580 | 4,5,6,7-Tetrahydro-1H-benzoimidazole-5-carboxylic acid New | 26751-24-6 |
An impurity of Ramosetron.Ramosetron is a serotonin 5-HT3 receptor antagonist for the treatment of nausea and vomiting.
|
|||
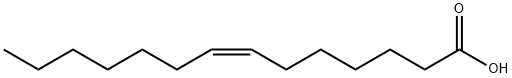 |
BCP49569 | (Z)-Tetradec-7-enoic acid New | 2430-95-7 |
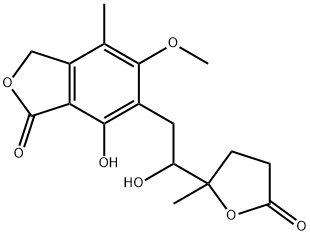 |
BCP49544 | Mycophenolate Hydroxy Lactone New | 26644-06-4 |
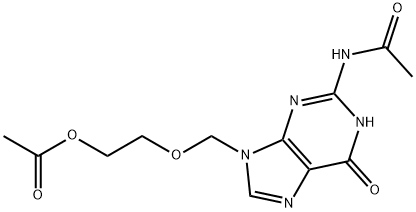 |
BCP26967 | Diacetylacyclovir New | 75128-73-3 |
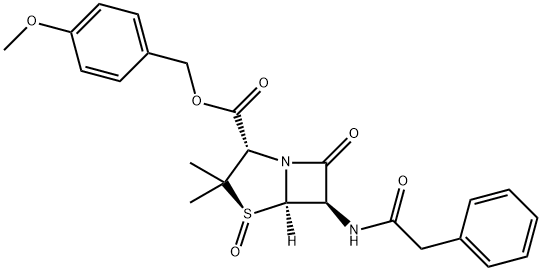 |
BCP27278 | Penicillin G sulfoxide P-methoxybenzyl ester New | 30034-13-0 |
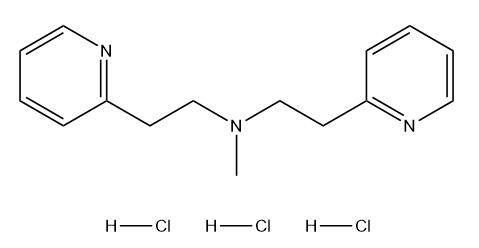 |
BCP49139 | Betahistine EP Impurity C Trihydrochloride New | 2095467-43-7 |